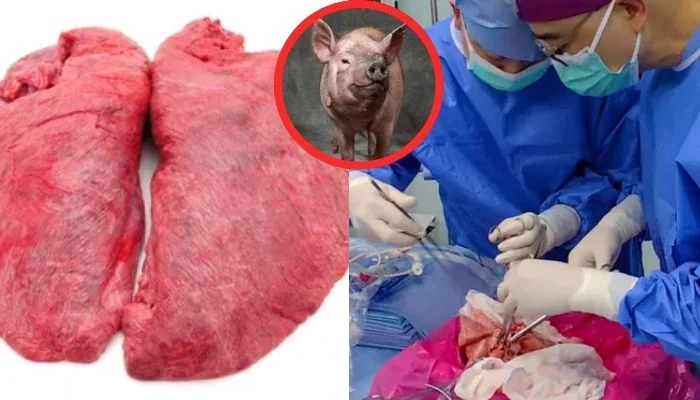
Genetically Modified Pig Lung Functions for Nine Days in Groundbreaking Transplant
In a historic medical achievement, Chinese surgeons have successfully transplanted a genetically modified pig lung into a human body, marking the first recorded case of its kind. The lung remained viable and fully functional for nine days in the recipient’s body without any signs of infection or rejection, according to a study published in Nature on Tuesday.
The recipient was a 39-year-old brain-dead man, making the procedure part of a pre-clinical trial to explore future applications in living patients. The lung underwent six genetic modifications to improve compatibility with the human immune system, a major hurdle in xenotransplantation—the use of animal organs in humans.
“Lungs are one of the most difficult organs to transplant due to their fragility and constant exposure to air,” noted Beatriz Dominguez-Gil, director of the National Transplant Organisation. She called the breakthrough a “milestone in translational medicine,” potentially paving the way for future lung transplants in living patients.
The study’s corresponding author, He Jianxing, told China’s Xinhua News Agency that the successful operation demonstrates critical progress in addressing global organ shortages. “This is a vital step forward in lung xenotransplantation,” he said, adding that the global demand for transplantable organs far outpaces available human donors.
Hope for the Future as Pig Organs Gain Ground in Transplant Science
The operation is part of a broader wave of experimental xenotransplantation procedures. To date, at least six individuals in China and the United States have received genetically engineered pig organs—including hearts, kidneys, livers, and a thymus—as researchers test the boundaries of organ compatibility between species.
Dr. Muhammad Mohiuddin, a xenotransplantation pioneer at the University of Maryland who led the first successful pig-heart transplant into a living human in 2022, praised the Chinese team. “Lungs are the most challenging organ to transplant,” he told Nature. “I applaud their effort. It’s a first step.”
The lung used in the transplant was specially bred and edited to reduce the risk of hyperacute immune rejection, a common problem when foreign tissues are introduced into the human body. The absence of infection or rejection over the nine-day observation period is a strong indicator that such transplants may become more viable in clinical settings.
While the patient was not alive at the time of the transplant, scientists say the next phase will involve limited trials in living humans, with extreme care to monitor long-term safety and efficacy.
The breakthrough is expected to accelerate global interest in genetically modified organ research, as the medical community continues its search for sustainable solutions to critical shortages in donor organs.